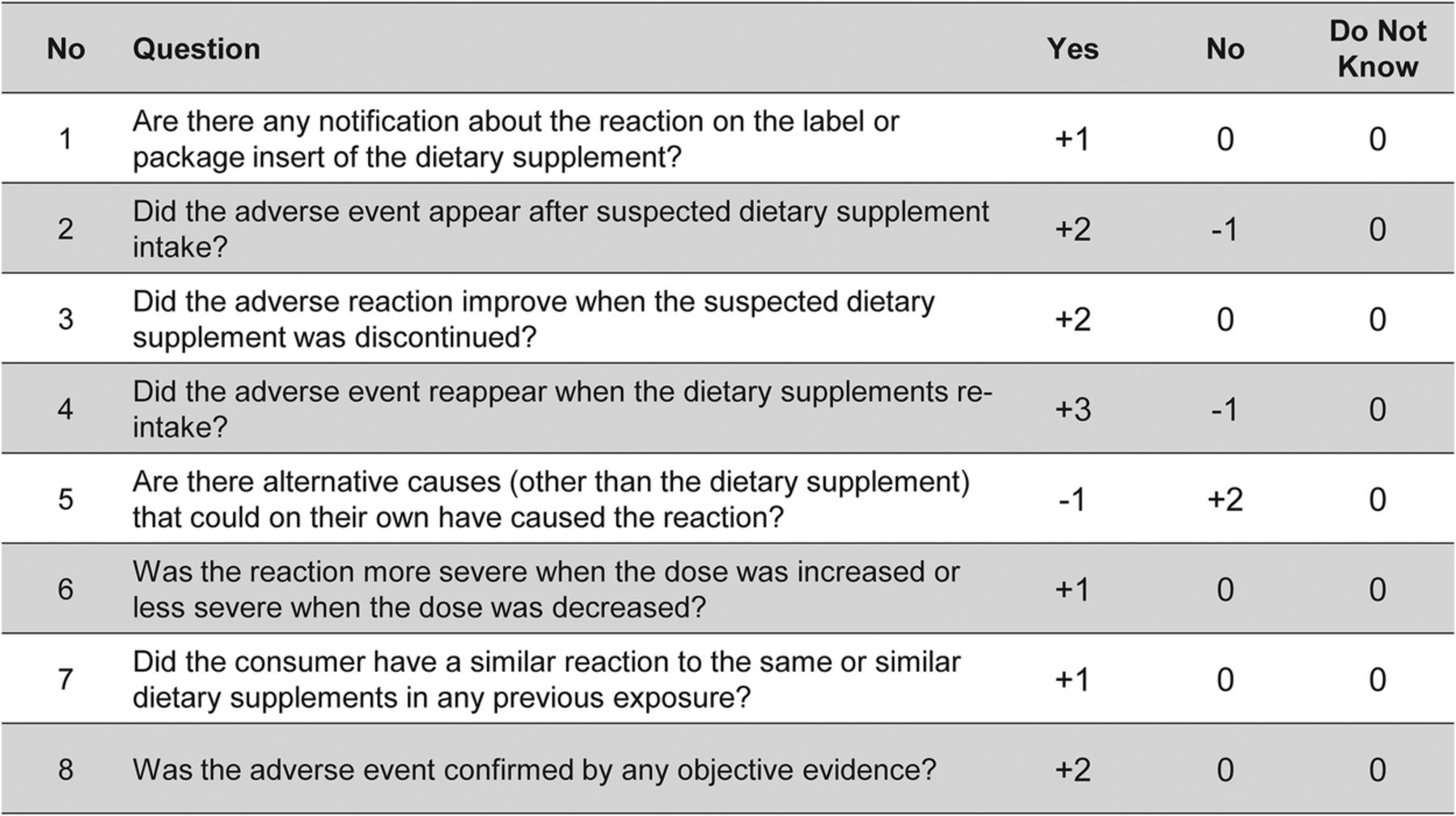
Causality assessment of adverse drug reactions: comparison of the results obtained from published decisional algorithms and from the evaluations of an expert panel. Macedo AF, Marques FB, Ribeiro CF, et al. The use of the WHO-UMC system for standardized case causality assessment.

World Health Organization (WHO), Uppsala Monitoring Centre. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. Reporting of fatal adverse drug reactions. Drug-related emergency department visits and hospital admissions. Admissions caused by adverse drug events to internal medicine and emergency departments in hospitals: a longitudinal population-based study. Schneeweiss S, Hasford J, Gottler M, et al. In conclusion, there is still no method universally accepted for causality assessment of ADRs. Because assessment methods are also not entirely devoid of individual judgements, inter-rater reliability can be low. Different causality categories are adopted in each method, and the categories are assessed using different criteria. As a result of problems of reproducibility and validity, no single method is universally accepted. The prior probability is calculated from epidemiological information and the posterior probability combines this background information with the evidence in the individual case to come up with an estimate of causation. Bayesian approaches use specific findings in a case to transform the prior estimate of probability into a posterior estimate of probability of drug causation. Algorithms are sets of specific questions with associated scores for calculating the likelihood of a cause-effect relationship. Expert judgements are individual assessments based on previous knowledge and experience in the field using no standardized tool to arrive at conclusions regarding causality. Thirty-four different methods were found, falling into three broad categories: expert judgement/global introspection, algorithms and probabilistic methods (Bayesian approaches). We conducted electronic searches in MEDLINE (via PubMed), EMBASE and the Cochrane databases to find all assessment methods. The aim of this review is to provide an overview of these methods and discuss their strengths and weaknesses.

Examples of common drug-induced reactions in these organ systems are also provided to demonstrate the process of assessing such reactions.Numerous methods for causality assessment of adverse drug reactions (ADRs) have been published. In addition to an introduction to the methodology for evaluating causality, this chapter focuses on the evaluation of side effects seen in commonly affected organs/organ systems (the liver, kidney, skin, and cardiovascular system) and highlights the factors that may need to be considered within these areas to determine a causal relationship. The ability to discern if a side effect is truly caused by a specific drug or is confounded by the patient’s concurrent medical condition(s) and/or concomitant medications is an art based on the totality of the available evidence combined with a systematic approach. Examples of common drug-induced reactions in these organ systems are also provided to demonstrate the process of assessing such reactions.ĪB - An adverse drug reaction is a side effect for which the cause can be directly attributed to a drug and its physiologic properties.

N2 - An adverse drug reaction is a side effect for which the cause can be directly attributed to a drug and its physiologic properties. T1 - Causality assessment and examples of adverse drug reactions (drug-induced liver injury, renal, skin, and major adverse cardiac events)
